Researchers measuring organic aerosol composition across multiple countries have found methylsiloxanes — silicone compounds widely used as engine oil additives — in air samples from urban centers, agricultural villages, and remote forest sites. The findings, published in Atmospheric Chemistry and Physics, raise new questions about an airborne contaminant that has received far less regulatory attention than microplastics or PFAS.
Combustion Survival and Long-Range Atmospheric Dispersion
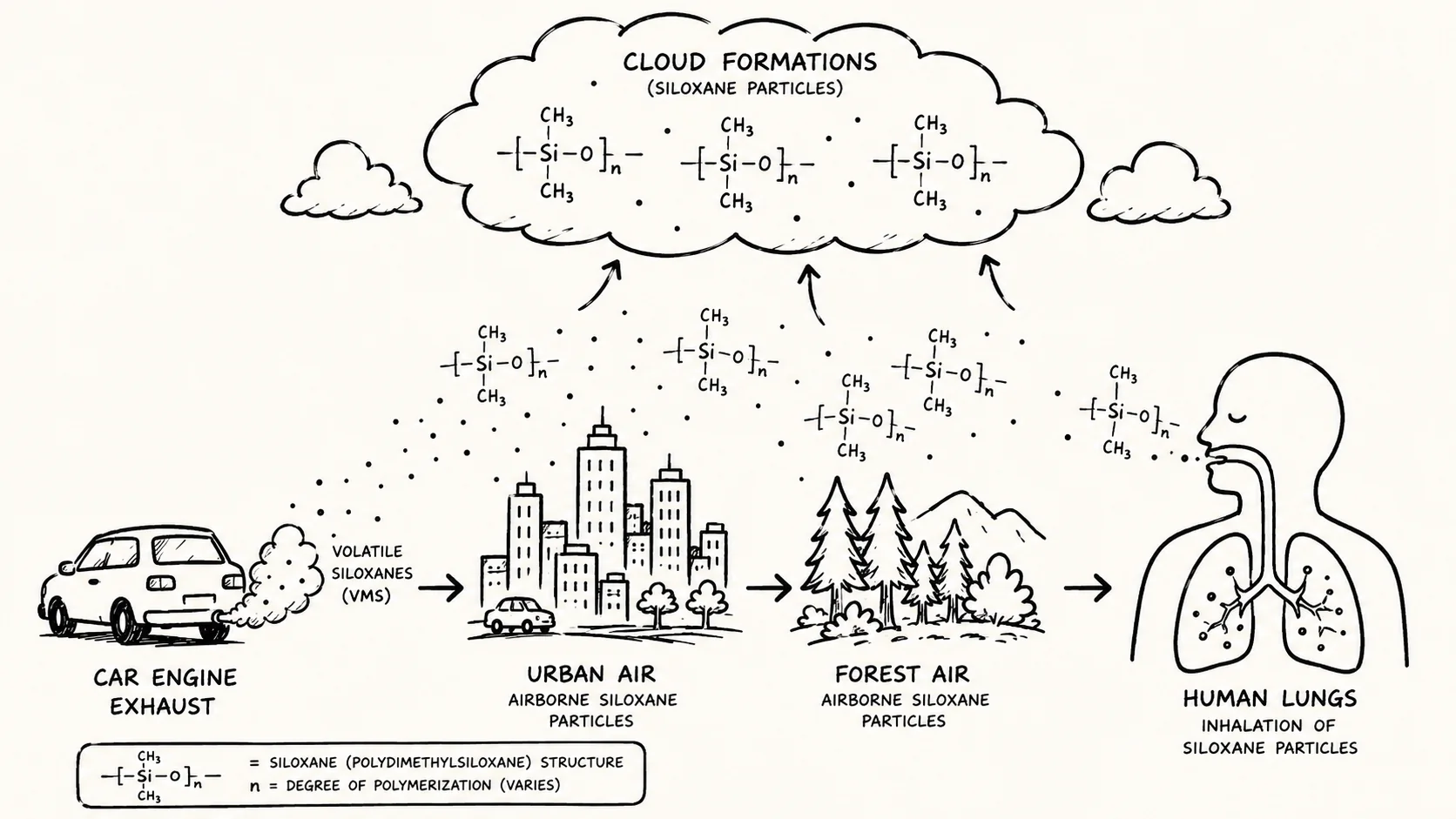
The pathway identified in the study is straightforward but the implication is not. Methylsiloxanes are added to engine lubricants because of their thermal stability — they resist breakdown at high temperatures. That same resistance means they survive the combustion process rather than being destroyed in the engine, escape through vehicle exhaust, and enter the atmosphere as stable particles. Unlike long-chain hydrocarbons from petroleum combustion, which fragment and dilute relatively quickly after emission, methylsiloxanes retain their molecular structure, allowing them to travel far from emission sources.
The researchers detected the compounds at sites that represent a wide range of human activity: São Paulo in Brazil, the village of Cabauw in the Netherlands, and a forest monitoring site in Lithuania. The presence of measurable concentrations in a Lithuanian forest suggests the compounds are not simply a localized urban problem. Their stability allows dispersion across regional distances.
The chart below maps the full pathway from engine oil to atmospheric exposure, as documented in the study.
Measured Concentrations Across Urban, Rural, and Forest Environments
The study provides specific concentration data across environments. At urban sites the researchers recorded peak concentrations of 98 nanograms per cubic meter. In the Lithuanian forest, the lower end of measurements reached 0.9 nanograms per cubic meter. Across all measurement sites, methylsiloxanes accounted for 2% to 4.3% of total organic aerosol mass — a share the researchers describe as unexpectedly high for a class of compounds that has not been a primary focus of air quality monitoring programs.
The comparison to PFAS and microplastics is worth treating carefully. The researchers estimated that people may inhale more methylsiloxanes on a daily basis than either of those two well-publicized contaminant classes. That estimate reflects the quantities moving through air, not a finding about equivalent biological effect. PFAS and microplastics have documented accumulation and toxicity profiles built on years of study. Methylsiloxane inhalation at these concentrations has no equivalent body of long-term evidence yet, and the researchers identified that gap explicitly as requiring urgent evaluation.
What distinguishes methylsiloxanes from conventional hydrocarbon combustion byproducts is not reactivity but persistence. Longer-chain hydrocarbons that escape combustion typically break down or dilute relatively quickly in the lower atmosphere. Methylsiloxanes do not. Their thermal and chemical stability — the same property that makes them useful as lubricant additives — means they can persist long enough in the atmosphere to travel from emission sources to remote environments, as the Lithuania data suggests. This is the property that gives the aerosol mass fraction finding its significance: it is not a local exhaust plume artifact; it is a broadly distributed background level. The following chart shows how measured concentrations varied across the three environment types documented in the study.
What the Study Does and Does Not Establish About Health and Climate Risk
The finding that these compounds reach remote forest environments at measurable concentrations is new and significant as a distributional observation. The study documents their presence, characterizes their source, and identifies two downstream concerns — human inhalation and atmospheric effects — without resolving either.
On the health side, the inhalation comparison to PFAS and microplastics is a quantity estimate, not a toxicological equivalence. PFAS compounds carry well-documented health associations from decades of epidemiological and laboratory work. Methylsiloxane inhalation at the concentrations now documented in ambient air has not been studied over equivalent timescales or in equivalent populations. The researchers are explicit that long-term health consequences are unknown and require urgent evaluation. That gap is not a minor caveat; it means there is currently no basis for characterizing the risk, only for recognizing that exposure exists at a broader scale than previously understood.
On the climate side, the paper describes two physical mechanisms: methylsiloxanes can alter aerosol surface tension and can interfere with ice nucleation, the process by which water vapor crystallizes around particles to form clouds. Both effects, if confirmed at scale, would have implications for how clouds form and how much solar radiation they reflect. Again, the study identifies the mechanisms and the presence of the compounds; it does not quantify net climate forcing. The pathway from measured atmospheric concentration to modeled climate effect involves additional steps that this study does not complete.
What the research does establish clearly is that a compound class defined by chemical stability — engineered to remain intact under extreme engine conditions — behaves with that same stability once it enters the atmosphere. That property warrants attention from both air quality regulators and atmospheric modelers who have focused monitoring infrastructure primarily on other pollutant categories.
Comments (1)
Please sign in to join the discussion.
We’ve spent years focusing on PM2.5, microplastics, and PFAS, but studies like this suggest there are still major airborne pollutants affecting human health that remain under-discussed and under-regulated. The fact that these particles can persist in the atmosphere and potentially interfere with both respiratory health and climate systems makes this issue even more concerning.
Hopefully this research pushes regulators and health agencies to investigate long-term inhalation effects more seriously.